A Randomized, Double-Blind, 3-Period Crossover, Pharmacokinetic Study in Healthy Adults to Compare Combined Kava and Kratom Supplementation with Kava or Kratom Alone Under Fasted Conditions
The study will consist of a total of 16 visits to our clinic (located in Guelph, Ontario) and 4 remote phone calls over a 22-week period. If you complete the study in its entirety, compensation will be up to $3,550 for your time and travel to the clinic.
The purpose of this study is to assess the pharmacokinetic (PK) properties of combined Kava and Kratom compared to Kava and Kratom individually in healthy adults.
Only a limited amount of spots available.
You may be eligible if you:
Are between 21 to 55 years old

Have a BMI between 18.5 to 29.9 kg/m²

Are in good general health
Do not regularly use tobacco, nicotine, or cannabis

Are not taking any prescription medications (including hormonal birth control)
Crossover Kava and Kratom Study
Participation will include:- 16 visits to our clinic (located in Guelph, ON) and 4 remote phone calls over a 22-week period
- 6 in-clinic visits lasting up to 16 hours each
- Consuming a liquid herbal supplement containing Kava root extract and/or Kratom leaf powder once daily with food during the treatment periods
- Multiple blood draws and urine sample collections
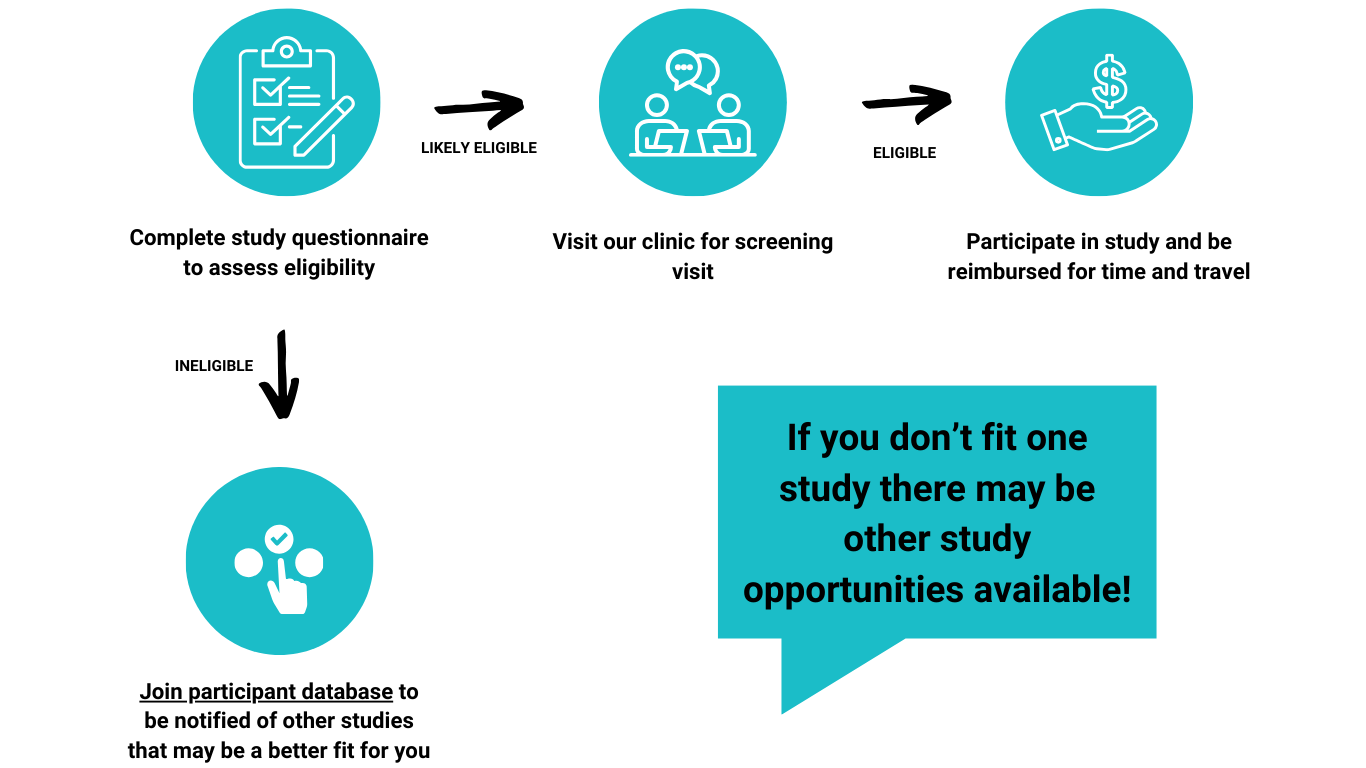
How It Works:
Facility Features
We take pride in our bright, modern clinic that we continue to upgrade to ensure a comfortable clinical trial experience for you. Click here to take a virtual tour of our space.

Free wifi available throughout facility

Accessible facility

Private rooms available for calls

Certified, experienced and friendly staff

Comfortable seating options

24/7 access to staff
The Team Behind The Scenes
At Apex Trials, your safety is our top priority. Our team of qualified staff are here to ensure everything runs smoothly.

Dr. Anthony Bier
Medical Director

Katie Keene
Clinic Manager

Ann Anton
Clinical Research Coordinator III

Chloe Gomes
Clinical Research Coordinator I
 Skip to main content
Skip to main content
